Vancouver, BC — June 27, 2022 — Telescope Innovations Corp. (“Telescope” or the “Company”) (CSE: TELI), a chemical technology company, announces the creation of “Telescope Automation”, a division of the Company that develops automated systems to accelerate chemical and biopharmaceutical research and development. Telescope Automation is pleased to launch its first product, the Direct Inject Liquid Chromatography (“DILC”) platform, an innovative instrument for real-time, immediate analysis of chemical reactions. The DILC platform fuses Telescope’s proprietary hardware, software, and unique integration know-how with proven sampling technology supplied by an internationally renowned manufacturer of lab instrumentation (the “Sampler Manufacturer”). The Sampler Manufacturer is the global leader in enabling hardware and software technology that accelerates the development of chemical and biological processes.
“We have been engaged by several major global pharmaceutical companies to onboard our DILC platform,” said Jason Hein, Telescope CEO. “Telescope has already leveraged this enabling tool across all our operations, and we are now bringing this to market as an unrivaled capability to address an unmet need in process analytics. The versatility and robustness of our platform surpasses other existing solutions for online chemistry analysis.”
This product launch enables Telescope to capture the fast-developing market opportunity for the DILC system, generate revenue, and establish the Company as a partner of choice for process laboratory tool integration. The commercial deployment and insight provided by DILC technology will highlight, at an industrial scale, the unique combination of skills Telescope provides to support chemical, biopharmaceutical, and manufacturing process development.
About Telescope’s Direct Inject (DILC) Platform
The DILC platform enables rapid, robust online analysis of chemical reactions. Specifically, the system samples batch or continuous processes and automatically quenches, dilutes, and injects the samples into a high- or ultra-performance liquid chromatography (“HPLC” or “UPLC”) instrument. When combined with orthogonal techniques such as spectroscopic analysis, this system provides full, comprehensive profiles of chemical reactions in real time (Figure 1).
The DILC system is fully automated and uses the market leading in-situ sampling probe (sold by the Sampler Manufacturer) to deliver reaction samples to HPLC/UPLC instruments in pseudo-real time. The entire process is controlled through Telescope’s intuitive software for workflow management. The DILC system ensures precise and reproducible samples, especially in heterogeneous and multiphase reactions or reactions at sub-ambient or elevated temperatures and pressures. Furthermore the DILC system eliminates delays in quenching that typically lead to variable results and inaccurate analytical information.
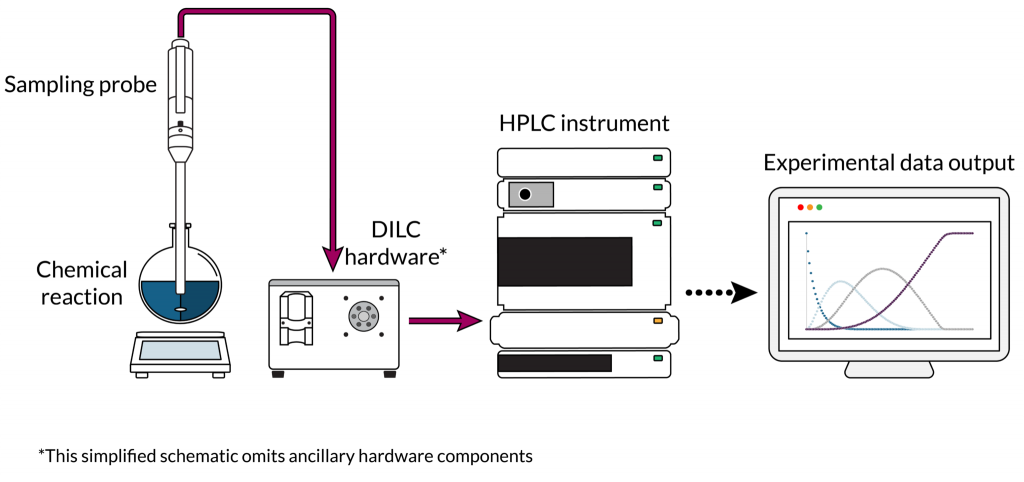
Figure 1. Simplified schematic of Telescope’s DILC system in an online chemical analysis setup.
Telescope’s DILC system provides many advantages in chemical process analysis:
- Rapid sampling frequency (the time period between sampling is near-HPLC method run times)
- No sample aging (sample is delivered directly to analysis with no exposure to the external environment)
- Adjustable dilution supporting a wide range of reaction concentrations
- Compatibility with reactions involving:
- biphasic liquid phases
- Daponte, J. A.; Guo, Y.; Ruck, R. T.; Hein, J. E.* “Using an automated monitoring platform for investigations of biphasic reactions.” ACS Catal., 2019, 9(12), p. 11484–11491.
- heterogeneous solid and liquid phases
- Sato, Y.; Liu, J.; Kukor, A. J.; Culhane, J. C.; Tucker, J. L.; Kucera, D. J.; Cochran, B. M.; Hein, J. E.* “Real-time monitoring of solid-liquid slurries: Optimized synthesis of tetrabenazine.” J. Org. Chem., 2021, 86(20), p. 14069-14078.
- Kukor, A. J.; Guy, M. A.; Hawkins, J. M.; Hein, J. E.* “A robust new tool for online solution-phase sampling of crystallizations.” React. Chem. & Eng., 2021, 6(11), p. 2042-2049.
- both liquid and gas phases
- Epifanov, M.; Foth, P. J.; Gu ,F.; Barrillon, C.; Kanani, S. S.; Higman, C. S.; Hein, J. E.; Sammis, G. M.* “One-Pot 1,1-Dihydrofluoroalkylation of amines using sulfuryl fluoride.” J. Am. Chem. Soc., 2018, 140(48), p. 16464-16468.
- reactions within an inert environment using air- and/or water-sensitive reagents
- Malig, T. C.; Yunker, L. P. E.; Steiner, S.; Hein, J. E.* “Online High-Performance Liquid Chromatography (HPLC) analysis of Buchwald-Hartwig aminations from within an Inert environment.” ACS Cat., 2020, 10(22), p. 13236-13244.
- Deem, M. C.; Derasp, J. S.; Malig, T. C.; Legard, K.; Berlinguette, C. P.; Hein, J. E.* “Ring walking as a regioselectivity control element in Pd-catalyzed C-N cross-coupling.” Nature Commun., 2022, 13, 2869.
- biphasic liquid phases
About Telescope
Telescope is a chemical technology company developing scalable manufacturing processes and tools for the pharmaceutical and chemical industry. The Company builds and deploys new enabling technologies including flexible robotic platforms and artificial intelligence software that improves experimental throughput, efficiency, and data quality. A key area of application for these tools is the development of scalable manufacturing processes for mental health medicines in the under-utilized tryptamine class of compounds, including psychedelic therapeutics. Telescope also applies these toolsets to resolve inefficiencies in industrial process chemistry and manufacturing. Our aim is to bring modern chemical solutions to meet the most serious challenges in health and sustainability.
On behalf of the Board,
Telescope Innovations Corp.

Jason Hein, Chief Executive Officer
Forward-Looking Information
Forward-looking information is necessarily based on a number of opinions, assumptions and estimates that, while considered reasonable by the Company as of the date of this news release, are subject to known and unknown risks, uncertainties, assumptions and other factors that may cause the actual results, level of activity, performance or achievements to be materially different from those expressed or implied by such forward-looking information, including but not limited to the factors described in greater detail in the “Risk Factors” section of the prospectus filed by the Company and available at www.sedar.com.
Forward-looking statements in this document include expectations surrounding the intended operation and use of the DILC platform, the ability of the DILC platform to generate revenue and the commercial viability of marketing the DILC platform to existing pharmaceutical companies, and all other statements that are not statements of historical fact.
Examples of such assumptions, risks and uncertainties include, without limitation, assumptions, risks and uncertainties associated with the global COVID-19 pandemic, including the risk that the Company be deemed a non-essential business and asked to temporarily cease operations; general economic conditions; adverse industry events; the willingness of existing pharmaceutical companies to adopt and utilize the DILC platform; the Company’s ability to access sufficient capital from internal and external sources, and/or inability to access sufficient capital on favorable terms; the ability of the Company to implement its business strategies; competition; and other assumptions, risks and uncertainties.
These factors are not intended to represent a complete list of the factors that could affect the Company; however, these factors should be considered carefully. There can be no assurance that such estimates and assumptions will prove to be correct. The forward-looking statements contained in this news release are made as of the date of this news release, and the Company expressly disclaims any obligation to update or alter statements containing any forward-looking information, or the factors or assumptions underlying them, whether as a result of new information, future events or otherwise, except as required by law.
The CSE has neither approved nor disapproved the contents of this news release. Neither the CSE nor its Market Regulator (as that term is defined in the policies of the CSE) accepts responsibility for the adequacy or accuracy of this release.

