Vancouver, BC — March 1, 2022 — Telescope Innovations Corp. (“Telescope” or the “Company”) (CSE: TELI), a chemical technology company, is pleased to provide a corporate update summarizing the significant milestones the Company has achieved over the past year of operations. The rapidly growing Company has delivered on the following three milestones that it believes set the foundation for unlocking future value for shareholders:
- Established a growing intellectual property portfolio on pharmaceutical manufacturing,
- Increased in-house research capacity and proprietary research platforms, and;
- Brought in award winning scientific leadership.
“In a little over a year, we have established Telescope as a deep tech company applying innovative chemistry technology to challenges in the pharmaceutical industry.” Said Jason Hein, CEO of Telescope. “We have discovered novel, scalable routes to manufacture psilocybin and address a huge unmet need in the treatment of mental health. Beyond this application, we are developing a robust set of technology platforms that accelerate chemistry research by leveraging robotic automation and artificial intelligence. We look forward to building out from this core foundation and deploying our systems to meet the biggest challenges in healthcare and chemical manufacturing.”
1. Established a growing intellectual property portfolio valuable for pharmaceutical manufacturing
One of Telescope’s strategic aims is to capitalize on the demand for the pharmaceutical-grade psychedelic therapeutics that are poised to address a growing US$ 16 billion market in anxiety disorders and depression treatment. While demand is growing, there is currently no regulated safe supply of psilocybin for therapeutic use. The Company has therefore focused on developing proprietary, scalable synthetic processes compatible with current good manufacturing practices (“cGMP”), with a view to sell and/or license this intellectual property to pharmaceutical companies or therapeutic providers.
Telescope has built out an intellectual property portfolio that currently consists of synthetic routes for the scalable production of pharmaceutical-grade psilocybin and psilocin (the active chemicals in psychedelic mushrooms) as well as their molecular precursors, analogs, and derivatives. These routes, in addition to a group of novel chemical entities, are captured in a Patent Cooperation Treaty Application (“PCT”) filed by Telescope in December 2021.
Telescope’s proprietary chemical manufacturing methods have several key advantages (Figure 1):
- They require fewer synthetic steps than other available methods, reducing supply chain costs, waste streams, and investments in the isolation and purification of intermediate compounds. Shorter manufacturing cycles also accelerate deployment and help maintain a competitive position.
- Unlike existing synthetic methods, we avoid the use of highly flammable chemicals that are prohibitively hazardous at a large scale.
- We combine chemical manufacturingtechniques that are standard practice in the pharmaceutical industry. This feature lowers the development costs to deploy our synthetic route and enables faster profitability.
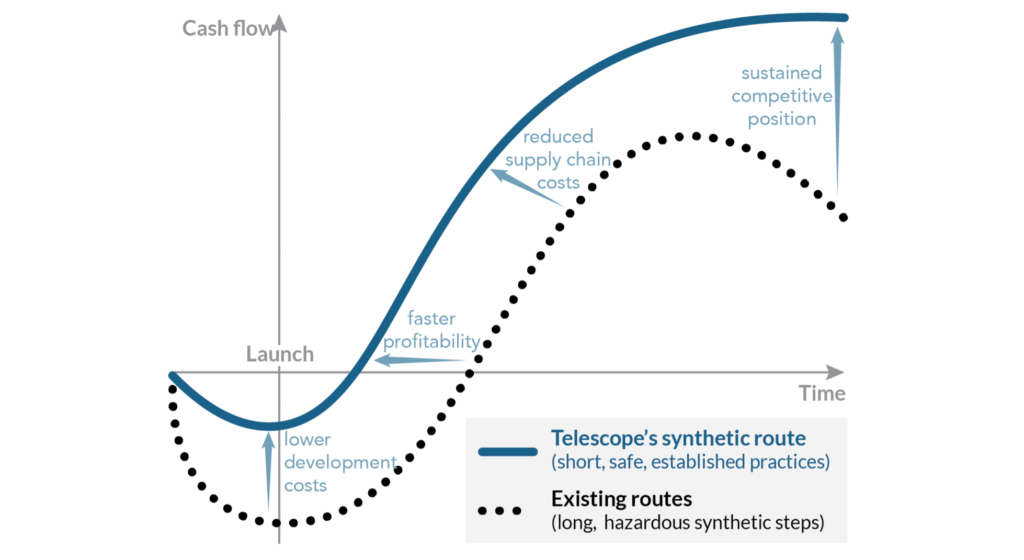
Figure 1. Short, safe synthetic processes using established methods (such as Telescope’s proprietary routes to psilocybin) provide significant economic benefits for commercial manufacturing.
- Our manufacturing routes are purposely flexible and easily tunable to produce valuable compounds in the same chemical family (e.g., dimethyltryptamine, harmaline, miprocin, ibogaine, melatonin, lysergic acid diethylamide, serotonin, bufotenine, etc.). The routes can also be tailored to produce novel molecules that may increase the safety, potency, or bioactivity of new psychedelic therapeutics. Even if a specific chemical compound fails to achieve FDA approval or pass regulatory milestones, Telescope’s manufacturing route can rapidly pivot to target the next generation of psychedelic drug candidates.
Within the PCT application filed in December 2021, Telescope has also captured a set of novel chemical entities (“NCEs”) to build a proprietary library of psychoactive compounds. The newly discovered molecules may perform similarly or more efficiently than psilocybin or other psychedelic medicines, and may become candidates for preclinical development as next-generation therapeutics in the future (Figure 2). NCEs may display improvements in safety, potency, specificity, or tunability, for example. To evaluate this potential, Telescope will pursue biological testing of these proprietary compounds by third-party, world-leading neurochemistry experts. Initial testing will aim to establish the effectiveness of the NCEs relative to leading psychoactive candidates.
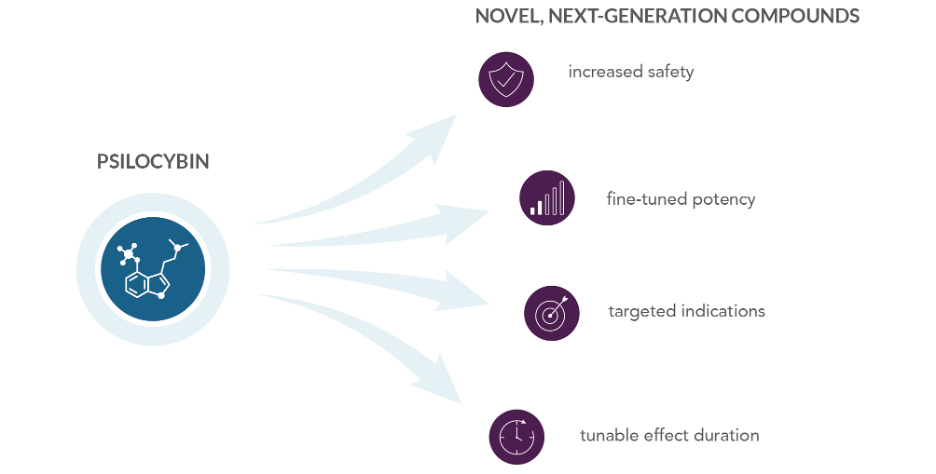
Figure 2. Telescope’s intellectual property portfolio includes a set of novel chemical entities in the same chemical family as psilocybin. These new compounds may pave the way for accessing the next generation of psychedelic therapeutics.
Telescope plans to expand this intellectual property moat over the next year to increase the scope of proprietary chemical reactivity and accessible products. The Company is also building relationships with biotechnology and pharmaceutical companies to explore avenues for monetization.
2. Increased in-house research capacity and proprietary research platforms
As of June 2021, Telescope occupies a dedicated laboratory facility in Vancouver, British Columbia. Here, we are developing and deploying our unique, proprietary ReACT platforms (Figures 3 and 4): robotic systems guided by artificial intelligence that increase the efficiency and quality of chemical research. We combine and integrate automation systems into a next-generation, self-guided chemistry experimentation platform that provides rapid, reliable data. This approach to research has enabled us to develop Telescope’s synthetic manufacturing processes for psilocybin in under one year.
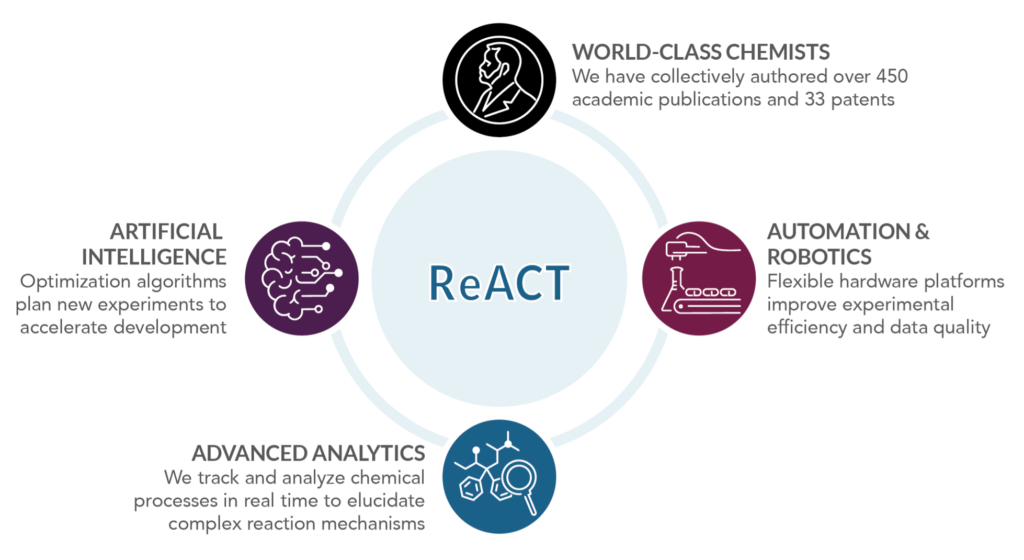
Figure 3. Telescope’s ReACT platforms dramatically accelerate chemistry research.
ReACT is powered by our cross-disciplinary technical team of organic, process, and analytical chemists, computer scientists, and mechatronics engineers. The seamless integration of hardware, software, and chemistry knowledge is key to rapidly advancing Telescope’s technical goals.

Figure 4. A robotic arm module used in Telescope’s ReACT platforms.
Telescope’s high-level aim is to transform industrial research and development by deploying its automated platforms and chemical technology across the pharmaceutical and chemical manufacturing sectors. Over the next quarter, we aim to engage in consulting agreements and research contracts with specific, major global pharmaceutical and chemical manufacturing companies to apply our research platforms in industrial settings. These projects would generate near term cash flow while validating Telescope’s ability to break down process barriers and increase the efficiency of industrial R&D. We are also exploring relationships with global laboratory instrumentation and automation companies to commercialize Telescope’s automation platforms, aiming to manufacture, market, and sell these platforms over the next two years.
3. Brought in award-winning scientific leadership
Telescope’s technical leadership comprises internationally renowned experts in chemical manufacturing and automation, having collectively authored over 450 academic publications and 33 patents.
- Prof. Barry Sharpless (Senior technical advisor) received the Nobel Prize in Chemistry in 2001 for “the most important discovery in the field of synthesis during the past few decades”. He received the Priestley Medal (the highest honour in the American Chemical Society) in 2019 and is the W.M. Keck Professor of Chemistry at the Scripps Research Institute in La Jolla, California.
- Prof. Jason Hein (CEO, Director) leads one of the largest academic chemistry research groups in Canada at the University of British Columbia. Has authored 50+ peer-reviewed articles amassing over 4,000 citations on organic chemistry, commercial manufacturing, and the role of automation, robotics and artificial intelligence in controlling pharmaceutical production.
About Telescope
Telescope is a chemical technology company developing scalable manufacturing processes and tools for the pharmaceutical and chemical industry. The Company builds and deploys new enabling technologies including flexible robotic platforms and artificial intelligence software that improves experimental throughput, efficiency, and data quality. A key area of application for these tools is the development of scalable manufacturing processes for mental health medicines in the under-utilized tryptamine class of compounds, including psychedelic therapeutics. Our aim is to bring modern chemical solutions to meet the most serious challenges in human health.
On behalf of the Board,
Telescope Innovations Corp.
Jason Hein, Chief Executive Officer
Forward-Looking Information
Forward-looking information is necessarily based on a number of opinions, assumptions and estimates that, while considered reasonable by the Company as of the date of this news release, are subject to known and unknown risks, uncertainties, assumptions and other factors that may cause the actual results, level of activity, performance or achievements to be materially different from those expressed or implied by such forward-looking information, including but not limited to the factors described in greater detail in the “Risk Factors” section of the prospectus filed by the Company and available at www.sedar.com.
Forward-looking statements in this document include expectations surrounding revenue streams from the research agreements, the patent filing process and the timing thereof, uses and viability of the intellectual property portfolio held by the Company, the performance of novel chemical entities, anticipated trends in the demand for psychedelic medical treatments and the ability of the intellectual property portfolio of the Company to respond to this demand, and all other statements that are not statements of historical fact.
Examples of such assumptions, risks and uncertainties include, without limitation, assumptions, risks and uncertainties associated with the global COVID-19 pandemic, including the risk that the Company be deemed a non-essential business and asked to temporarily cease operations; general economic conditions; adverse industry events; future legislative and regulatory developments involving psilocybin; the Company’s ability to access sufficient capital from internal and external sources, and/or inability to access sufficient capital on favorable terms; the psilocybin industry in Canada and generally; the ability of the Company to implement its business strategies; competition; and other assumptions, risks and uncertainties.
These factors are not intended to represent a complete list of the factors that could affect the Company; however, these factors should be considered carefully. There can be no assurance that such estimates and assumptions will prove to be correct. The forward-looking statements contained in this news release are made as of the date of this news release, and the Company expressly disclaims any obligation to update or alter statements containing any forward-looking information, or the factors or assumptions underlying them, whether as a result of new information, future events or otherwise, except as required by law.
The CSE has neither approved nor disapproved the contents of this news release. Neither the CSE nor its Market Regulator (as that term is defined in the policies of the CSE) accepts responsibility for the adequacy or accuracy of this release.
